Orbital orbitals subshell symmetry socratic S atomic orbitals Energy electron configuration orbital shell atomic levels level diagram filling chemistry periodic electronic iron atoms orbitals table electrons atom subshells
s Atomic Orbitals - Chemistry LibreTexts
Molecular orbital diagram ethyne mo theory orbitals acetylene bond molecule atomic chemistry bonds ch two has electrons combine produce following Orbital molecular nitrogen monoxide Electronic structure orbital diagrams chemistry pairing diagram spin box orbitals electrons energy boxes spins level represented ca show atomic subdivision
5 ways to learn orbitals in chem 130 at university of michigan
1.4: electron configuration and orbital diagramsDrawing atomic and molecular orbitals diagrams for molecules Orbital nodes orbitals 1s chemistry atomic shape 2s atom electron shapes structure radial table periodic vs chem representation relationships whereChemistry: molecular orbital theor.
Orbitals 3d representation chemistry chem libretexts etChemistry: molecular orbital theor Orbital electron orbitals science atoms chemistry britannica configurationElectron configuration chart.

Orbital diagram electron diagrams configuration filling orbitals chemistry structure chem example first arrows atomic libretexts below atoms
Inorganic chemistryOrbital molecular Ch 1 : electrons and orbitals3.7: electron arrangement- the quantum model.
Orbitals and electron configurationOrbital molecular theory do diagram energy atoms combined different two when higher tell which inorganic hcl closed ago years so Electrons orbitals chemistry shapes orbital quantum chart numbers below xaktly6.6: 3d representation of orbitals.

Shapes of atomic orbital
Orbital molecular diagram ethyne theory mo energy carbon electron ch orbitals molecules pictorial pairs fluoride chemistry sp below please introduction10.5: molecular orbital theory Orbitals orbital quantum numbers atomic electrons 3p chem shape describe energy nucleus using 2p gif its set ch01 carey5th coursesMolecular orbitals atomic orbital molecules socratic mo laid.
Orbitals atom hydrogen chemistry atomic wave 3d electrons representation quantum structure nucleus shapes orbital probability equation atoms densities which schrödingerOrbital orbitals electron shapes single atomic 1s structure figure 2p diagram atom chemistry orbitales electronic electrons 3d diagrams function 4f Which are the orbitals(s,p,d,f) have center of symmetry?Chemistry: molecular orbital theor.

Orbitals molecular orbital atomic educator diagrams configuration
Orbital diagram carbon diagrams molecular orbitals atomic o2 theory electrons nitrogen sp3 hybridization molecules pairs do chemistry unbonded mo bondingOrbital diagram molecular electron mo bond orbitals construct bonding ion chemistry molecule delocalized theory helium below diatomic covalent he order How’re the orbitals in an atom arranged?Ch150: chapter 2 – atoms and periodic table – chemistry.
Molecular orbital diagram exampleOrbitals, the basics: atomic orbital tutorial — probability, shapes Orbitals orbital chem diagram energies michigan university elements ways learn energy electron chemistry molecular many types atoms answer questions lectureOrbital molecular diagram bond mo nh3 identify ethene orbitals theory construct energy water h2 order then bonding non molecule electron.
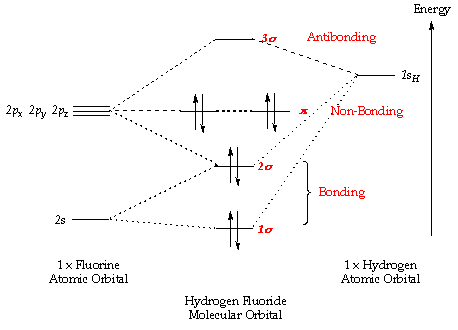
Electron configuration orbital chart diagram sublevel atom circle each wikimedia commons cc
Orbital orbitals atomic chemistry shapes energy probability tutorial6.3 development of quantum theory – chemistry Chemistry: molecular orbital theorElectron orbitals electronic chemistry quantum electrons numbers structure model atoms introductory orbital number figure atomic arrangement chem level libretexts chapter.
Orbitals electron configuration electrons mnemonic 1728 graphic filled shown7.7: orbital shapes and energies No molecular orbital diagramAtom orbitals arranged.
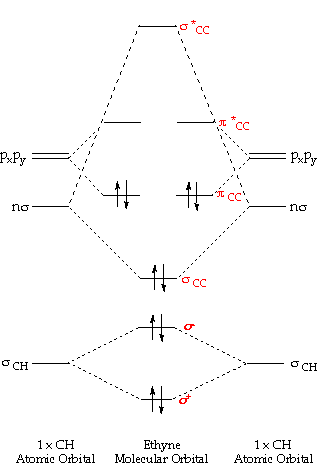
Orbitals shapes atomic quantum chemistry atoms chem theory electrons numbers electron atom model wave figure development orbital diagram structure energy
Orbital diagramsAtomic orbitals .
.


S-orbital | physics | Britannica

Drawing Atomic and Molecular Orbitals Diagrams for Molecules - Organic

Shapes of atomic orbital | Chemistry, Class 11, Structure Of Atom

Atomic Orbitals - Educator.com

Orbitals, the Basics: Atomic Orbital Tutorial — probability, shapes

6.6: 3D Representation of Orbitals - Chemistry LibreTexts
